Price 150 आईएनआर/ Box
MOQ : 500 Boxes
Specification
- Packaging Type
- Alu-Alu Blister
- Prescription/Non Prescription
- Prescription
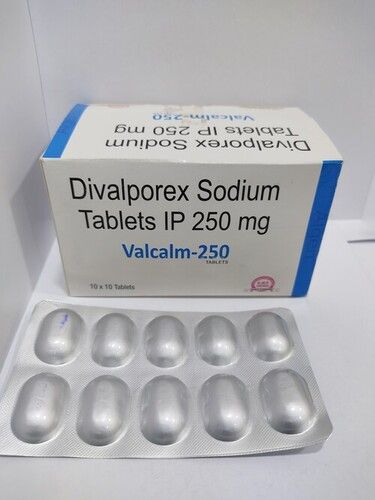
- Brand Name
- VALCALM-250
- Form
- Tablets
- Strength
- 250 mg
- Manufactured By
- Valience Healthcare Private Limited
- Therapeutic Class
- Antiepileptic
- Packaging Size
- 10x10 Tablets
- Storage Conditions
- Store in a cool, dry place below 25°C
- Marketed by
- Valience Healthcare Private Limited
- Usage/Application
- Clinical, Hospital
- Generic Name
- Divalproex Sodium Extended Release Tablets
- Indications
- Used for the treatment of seizure disorders, bipolar disorder, and migraine prophylaxis
Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
मोबाइल number
Email
 जांच भेजें
जांच भेजें
 जांच भेजें
जांच भेजें एसएमएस भेजें
एसएमएस भेजें मुझे निःशुल्क कॉल करें
मुझे निःशुल्क कॉल करें
